Phytoplankton Lab Studies Reveal Both Expected And Unexpected Results
Increasing CO2 in the air leads to more CO2 in the surface ocean home of the world’s most important plant life, the phytoplankton.
A little extra CO2 was expected to enhance phytoplankton growth as was observed but that increase was far offset by the suppression of growth and biomass as CO2 lowers ocean pH.
This work may help explain the 50% collapse of phyto-plankton in the ocean pastures surrounding Antarctica.
CO2 in world’s air is up by 40+% in just the past 100 years of the fossil fuel age. This CO2 has been producing global greening of plants on land and it seems in this, in vitro, test-tube study higher CO2 may also produce more plant growth of important ocean phytoplankton – Chaetoceros diatoms. The study however found a more disturbing role for CO2 was tied to light intensity. With maximum light the diatoms can indeed consume more CO2 and fight off the effects of their watery home turning acidic, and the oceans have become 30% more acidic from our fossil fuel CO2 emissions. But when the sunlight is at all diminished such as to levels found commonly now in nature the diatoms quickly succumb to the acid in their seawater. Even slight pH lowering as is now seen likely makes it energetically difficult for the diatoms to precipitate the dissolved minerals they need to thrive and survive.
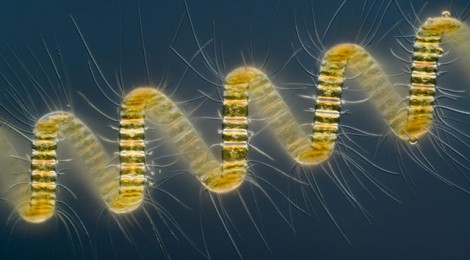
The big stony Chaetoceros diatoms, such as the one featured in the banner image of this post, are known by ocean scientists as the best carbon sinkers andas such they play a very critical role in ocean carbon ecology. Diatoms are phytoplanton that form a protective silicon carbonate shell that also serves as a means ballast allowing them to control their depth. They are a primary and major ocean pasture crop/food source at the very bottom of the food chain for all of ocean life. They are found worldwide but are especially abundant in polar waters. Antarctic krill in particular depend heavily on good crops of diatoms and in turn the penguins and great whales of the Antarctic depend on the krill.
But phytoplankton like the diatoms can get too much sunlight and UV and suffer ‘sunburn’ effects. When the day is especially bright they stay deeper beneath the protective sunscreen in more dimly lit water. Further phytoplankton also have the effect of making clouds, as their numbers reach bloom levels. The combined overall effect of our high and rising CO2 is terrible cloudy news for ocean pastures.

Alfred Wegner Institute and European Union Behemoth Ocean Research Flagship Polarstern in Antarctica is frequently tasked with collecting the very tiniest of life in that icy sea.
German ocean scientist Clara Hoppe and colleagues from the Alfred Wegener Institute in Bremerhaven, Germany, report in the journal New Phytologist that they tested the growth of the Antarctic diatom Chaetoceros debilis under laboratory conditions.
Hoppe and colleagues used two levels of pH – which is an indicator of CO2 driven acidity – and they exposed their diatom cultures to either constant intense maximum light of a perfect laboratory condition and they changed the light condition to one that simulated lower intensity light in the real world of the diatoms.

Diatoms growing in culture with artificial light in the laboratory. They can do ok here but it is a far different environment from the open ocean.
In the steady glare of perfect laboratory light, the diatoms responded as well as expected. Their growth levels were consistent with an assumption that more dissolved carbon dioxide – which makes the waters more acidic – would also stimulate extra diatom growth. Hurrah they proved diatoms are photosynthetic green plants!
However in the changing to lower light conditions found in nature the diatoms revealed a very different story. They grew more slowly, showing that these vital members of ocean pasture ecologies become less efficient at growing and repurposing CO2 into life that sustains the ocean pastures while more CO2 was left behind to become worsening ocean acidification.
“Diatoms fulfil an important role in the Earth’s climate system,” Dr Hoppe says. “They can absorb large quantities of carbon dioxide, which they bind before ultimately transporting part of it to the depths of the ocean. Once there, the greenhouse gas remains naturally sequestered for centuries.”
Previous research into the steady acidification of the oceans that has already dropped ocean pH by 30% has tended to concentrate on the consequences for coral reefs, fisheries, and tourism, but not on the impact on vital plant life in the ocean pastures far out to sea and away from the bathing beauties at the beach where, near beaches, the vast majority of ocean science is “performed.”
Dr Hoppe says: “Several times a day, winds and currents transport diatoms in the Southern Ocean from the uppermost water layer to the layers below, and then back to the surface – which means that, in the course of a day, the diatoms experience alternating phases with more and with less light.”
Her co-author, marine biogeochemist Björn Rost, from the Alfred Wegener Institute, says: “Our findings show for the first time that our old assumptions most likely fall short of the mark. We now know that when the light intensity constantly changes, the effect of ocean CO2 and its acidification action reverses.”
More CO2 in the water doesn’t mean more plant growth when one considers natural light conditions. In fact in the oceans natural environment more CO2 means less vital phytoplankton and that begins a terrible feedback cycle as phytoplankton are the only force in nature that competes for CO2 against the alternative and inevitable acidification chemistry.
Thus higher ocean CO2 and more acid/lower pH means fewer diatoms which mean even more acid in that terrible feedback cycle that hinges on the diatoms and their role as primary producer of ocean pastures being dealt an impossibly series of devastating blows. It’s already been established that something has been killing the phytoplankton and diatoms of the Southern Ocean as data has shown a horrific 50% decline in productivity of Southern ocean pastures in the past ~40 years. This work makes a good step in understanding just how this collapse of Southern Ocean plankton has gotten so bad.
One failing of this reported research is that it has serious limitations. It applies to one species of diatom in the waters of one ocean, and the tests were in a laboratory on a small-scale, and not in a turbulent ocean rich in life. The Alfred Wegener team will continue their studies.
Other real world evidence of CO2’s harmful effects are seen.
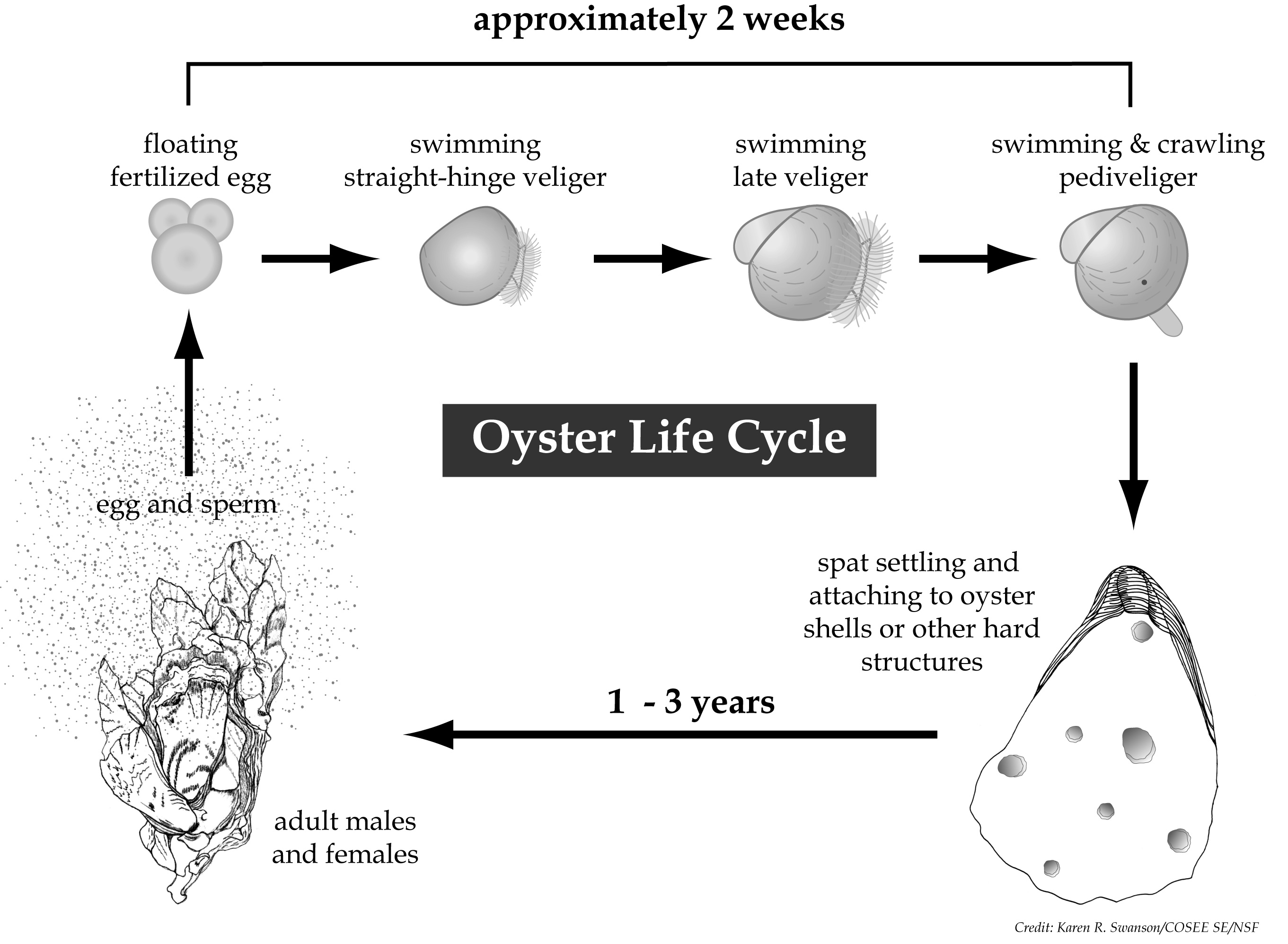
Oyster Early Life Cycle when they are most vulnerable to tiny changes in ocean pH (acidity)… when they have grown their first hair thin shell they sink to the bottom being just a tiny fraction of an inch long.
On the North American side of the Atlantic ocean scientists are reporting on a real world crisis of CO2 in the ocean that is already threatening ocean life, especially shellfish. A combination of high and rising CO2 levels in the air making ocean waters more acidic combined with upwelling currents bringing even more acidic deep ocean waters ashore is killing shellfish from the Pacific Northwest, New England, the Mid-Atlantic states and the Gulf of Mexico – affecting the shellfish industry that is worth at least $1 Billion each year in the USA.

Hair thin transparent shells on these baby oysters don’t have a chance of ever forming in oceans turning even weakly more acidic.
The key to understanding how CO2 driven changes in ocean pH effects shellfish is in knowing that before a big rock hard oyster, clam, or mussel grows it’s shell it must first start our life as a nearly microscopic larvae. Those larvae have little chance of ever-growing their first hair thin shell in oceans where the pH has been depressed as is the case today all over the world.
“Ocean acidification has already cost the oyster industry in the Pacific Northwest nearly $110 million and has jeopardized about 3,200 jobs,” Dr Ekstrom at the University of California at Davis says.